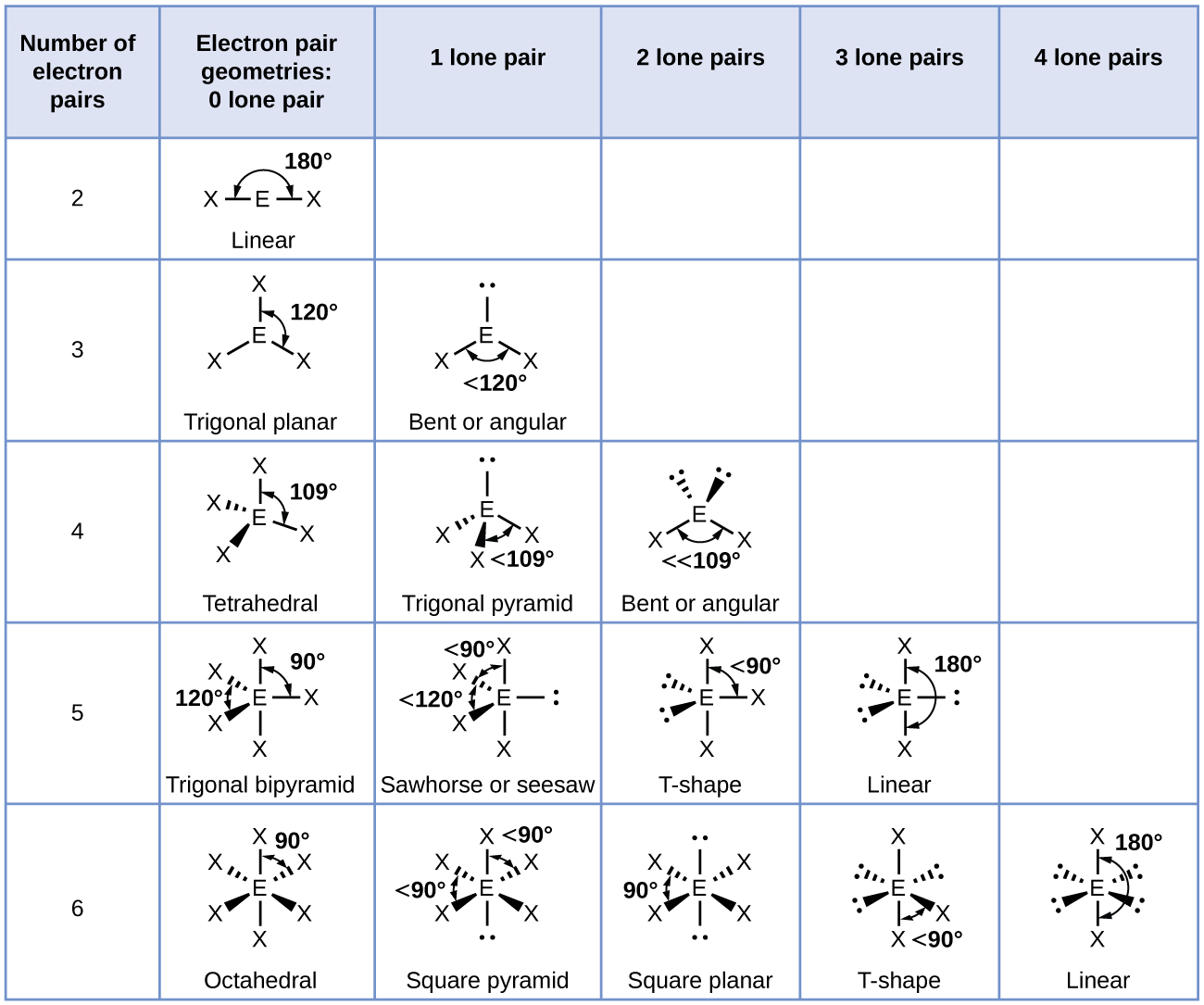
Now remember lone pairs have an electron cloud which further causes more repulsion. Now, another option that could occur is we have to surrounding elements or two bonding groups and one lone pair on the central element here we have our example with tin in the center with two chlorine and it has one lone pair in its visualization, we have lost the element that was here and it's been replaced by a long pair. It's visualization would look like this and we'd say that its molecular geometry will be tribunal player. Here we have an example of carbon in the center connected to three surrounding elements. In the first situation we have three surrounding elements or three bonding groups and zero lone pairs. So again, when we have three electron groups around our central element to possible shapes can occur. When it comes to three electron groups were going to say this is when central elements with three electron groups that can have either zero or one lone pair to get to possible molecular geometries. So when it comes to two electron groups around the central element, there's only one possible molecular geometry and that's a linear molecular geometry Hide transcripts The molecular geometry for all of them would be linear. Now it doesn't matter if it's single bonded to them or double bonded or triple bonded, it's still too surrounding elements or two bonding groups visually would see it as our black spear here, which is our central element connected to those gray spheres which are surrounding elements here. We have beryllium, carbon and carbon again, and they are connected to only two surrounding elements. Zero lone pairs here are some examples in all of them. Remember bonding groups are just your surrounding elements. So that means we're gonna have to bonding groups possible. So if we take a look here, We have two electron groups, we say that there are zero lone pairs. Now, when we have two electron groups are going to say here, this is central elements with two electron groups that have zero lone pairs and give only one possible molecular geometry. 
Now, we're going to take a look at each of the number of electron groups that exist. So they're not going to be treated as the same. Now, because of this we would treat lone pairs on the central element and surrounding elements as different. That takes into account differences and repulsion between lone pairs and surrounding elements. The molecular geometry of a compound can be seen as the true shape of that molecule compound.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
